An element having the atomic number 17 will resemble which of the following elements having atomic number as given below?
Options:A) 33
B) 35
C) 18
D) 19
Show Answer
The correct answer is B .
An element Z contains 80% of
A) 16.2
B) 17.6
C) 16.0
D) 16.4
Show Answer
The correct answer is D .
Helium is used in observation of ballons because it is?
Options:A) Light and combustible
B) light and non-combustible
C) heavy and combustible
D) heavy and non-combustible
Show Answer
The correct answer is B .
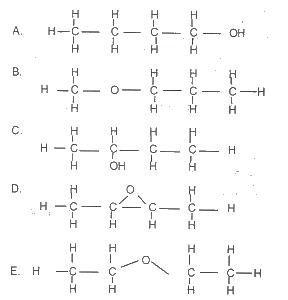
Use the diagram above to answer this question.
Which of the following structural formula is NOT isomeric with the others?
Options:A) A
B) B
C) C
D) D
E) E
Show Answer
The correct answer is D .
A) Low melting point
B) Weak electropositive character
C) High boiling point
D) White lustrous appearance
Show Answer
The correct answer is A .
A) 14
B) 13
C) 12
D) 11
Show Answer
The correct answer is D .
A) 4
B) 5
C) 2
D) 3
Show Answer
The correct answer is B .
A) xylene
B) toluene
C) cyclopentane
D) cyclohexane
Show Answer
The correct answer is D .
220g of carbon dioxide contains ----- moles
Options:A) 5
B) 6
C) 3
D) 4
Show Answer
The correct answer is A .
A) Na+ and Cl-
B) Na+ and OH-
C) H+ and OH-
D) H+ and Cl-
Show Answer
The correct answer is D .