A) Aluminium tetraoxosulphate (VI)
B) Copper tetraoxosulphate (VI)
C) Sodium tetraoxosulphate (VI)
D) Calcium tetraoxosulphate (VI)
Show Answer
The correct answer is A .
Which quantum divides shells into orbitals?
Options:A) Spin
B) Principal
C) Azimuthal
D) Magnetic
Show Answer
The correct answer is C .
A) strong intermolecular attraction
B) high melting point
C) high boiling point
D) weak intermolecular attraction
Show Answer
The correct answer is A .
A) 1 and 1
B) 1 and 2
C) 2 and 1
D) 3 and 1
Show Answer
The correct answer is C .
A) 2.0 mole of Q3+
B) 2.5 mole of R2+
C) 3.0 mole of T-
D) 4.0 mole of Y-
Show Answer
The correct answer is A .
A) Fe
B) Al
C) Zn
D) Pb
E) none of the options is correct
Show Answer
The correct answer is E .
A) potasium dioxonitrate (III)
B) lead (IV) oxide
C) potassium trioxochlorate (V)
D) potassium trioxonitrate (V)
Show Answer
The correct answer is A .
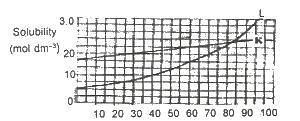
Use the solubility curves below to answer questions. If 1 dm3 of a saturated solution of L at 600C is cooled to 250C, what amount in mole will separate out?
Options:A) 0.75
B) 0.25
C) 1.00
D) 0.50
Show Answer
The correct answer is A .
A) more solute molecules collide with each other
B) most solutes dissolve with the evoltion of heat
C) more solute molecules dissociate at hihger temperatures
D) most solutes dissolve with absorption of heat
Show Answer
The correct answer is C .
A) positive change
B) negative entropy change
C) no entropy change
D) a minimum entropy change
Show Answer
The correct answer is C .