A) propane 1, 1, 3 - triol
B) propane - 1,2,3 - triol
C) propane - 1,3,3 - triol
D) propane - 1, 2, 2 - triol
Show Answer
The correct answer is B .
Deliquescent substances are also?
Options:A) efflorescent
B) anhydrous
C) hygroscopic
D) insoluble
Show Answer
The correct answer is C .
Which of the following factors will speed up the rate of evolution of carbon (iv) oxide in the reaction below?
2HCl + CaCO
A) Increasing the concentration of the acid
B) Increasing the surface area of CaCO
C) A and B
D) Reducing the concentration of the acid
Show Answer
The correct answer is C .
A) log 10[OH-]
B) log 101/[H 3 O+]
C) log 10[H 3 O+]
D) log 101/[OH -]
Show Answer
The correct answer is B .
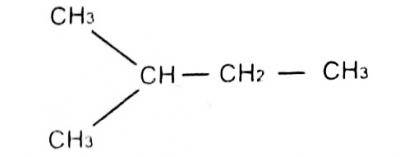
The lUPAC name for
Options:A) 1-methyl pentane
B) 3-methylbutane
C) 2-methylbutane
D) 1-dimethyl propane
Show Answer
The correct answer is C .
A) xylene
B) toluene
C) cyclopentane
D) cyclohexane
Show Answer
The correct answer is D .
The process of rusting is an example of the formation of
Options:A) Basic oxide
B) Acidic oxide
C) Reducing oxide
D) Amphoteric oxide
Show Answer
The correct answer is A .
A) negative
B) zero
C) positive
D) indeterminate
Show Answer
The correct answer is D .
Which of the following is a non-metal that remains liquid at room temperature?
Options:A) Chlorine
B) Phosphorus
C) Bromine
D) Helium
Show Answer
The correct answer is C .
Which of the following statements is true for strong electrolytes?
Options:A) They do not conduct electricity in the molten state
B) They completely dissociate into ions in solution
C) They are non-polar substances
D) They partially dissociate into ions in solution
Show Answer
The correct answer is B .