A) NaOH solution by 70 cm3
B) NaOH solution, by 60 cm3
C) NaOH solution by 40 cm3
D) Al(NO3)3 solution by 20 cm3
E) Al(NO3)3 solution by 10 cm3
Show Answer
The correct answer is C .
A) Helium
B) Magnesium
C) Copper pyrites
D) Glass
Show Answer
The correct answer is B .
Two equal bulbs, one containing ammonia and the other nitrogen are opened mouth-to-mouth to each other at room temperature. The entropy in the mixture of gases is likely to
Options:A) remain unchanged
B) increase
C) decrease
D) change
Show Answer
The correct answer is B .
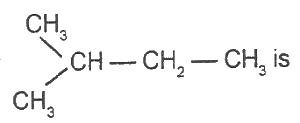
The IUPAC name for
Options:A) 1-methlpentane
B) 3-methylbutane
C) 2-methylbutane
D) 1-dimethylpropane
E) 2-methylpentane
Show Answer
The correct answer is C .
If one of the following oxides is heated with hydrogen or carbon using a bunsen burner. it is not reduced to the metal, Which one is it?
Options:A) lead oxide
B) Magnesium oxide
C) Copper oxide
D) Tin oxide
Show Answer
The correct answer is B .
A) sodium is formed at the anode
B) chlorine is formed at the anode
C) sodium does not react with platinum
D) chlorine does not react with platinum
Show Answer
The correct answer is C .
A) CO2 + C → 2CO
B) 2Fe2O3 + 3C → 4Fe + 3CO2
C) Fe2O3 + 3C → 2Fe + 3CO2
D) CaCO3 → CaO + CO2
E) CaO + SiO2 → CaSiO3
Show Answer
The correct answer is B .
Na
The indicator most suitable for this reaction should have a pH equal to
Options:A) 5
B) 7
C) 3
D) 9
Show Answer
The correct answer is C .
A) both gases change the colour of the solution from yellow to colourless
B) both gases causes sulphur to be deposited
C) only hydrogen sulphide decolourizes the solution
D) only sulphur dioxide deposits sulphur
E) only hydrogen sulphide deposits sulphur
Show Answer
The correct answer is E .
A) All the nitrate will decompose to their respective metals
B) The nitrates of calcium and potassium will decomposes to their nitrites
C) Only copper nitrate decomposes to the metal
D) Only the nitrates of zinc and copper will decompose to their oxides
E) The nitrates of calcium, zinc and copper decompose to their oxides
Show Answer
The correct answer is E .