A) pig iron
B) wrought iron
C) cast iron
D) iron pyrites
Show Answer
The correct answer is B .

In the diagram, the function of the concentrated H2SO4 is to
Options:A) purify the gas
B) dry the gas
C) liquefty the gas
D) remove odour
Show Answer
The correct answer is B .
A) oxidized to ethene
B) polymerized to polyethene
C) dehydrated to ethene
D) dehydrated to ethyne
Show Answer
The correct answer is C .
A) Fe
B) Al
C) Zn
D) Pb
E) none of the options is correct
Show Answer
The correct answer is E .
A) Distillation
B) Condensation
C) Extraction
D) Evaporation
Show Answer
The correct answer is A .
A) Chlorine
B) Sulphur (IV) oxide
C) Ammonia
D) Carbon (IV) oxide
Show Answer
The correct answer is C .
A) amino acids
B) glycerols
C) alkanols
D) alkanoates
Show Answer
The correct answer is B .
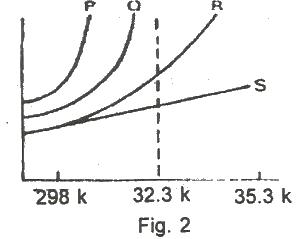
The solubility curves of four substances are shown in the figure above. Which of the four substances would crystallize from a saturated solution cooled from 353K(80oC)to 323K(50C)?
Options:A) P and Q
B) P and R
C) P and S
D) R and S
E) Q and R
Show Answer
The correct answer is D .
A) +7
B) +5
C) +3
D) +2
E) +1
Show Answer
The correct answer is A .
The reaction
A) not a redox reaction because there is no oxidant in the reaction
B) not a redox reaction because there is no reductant in the reaction
C) a redox reaction in which H
D) a redox reaction in which SO
Show Answer
The correct answer is D .