Addition of charcoal to the filter bed of sand during water treatment for township supply is to?
Options:A) prevent goiter
B) prevent tooth decay
C) remove odour
D) kill germs
Show Answer
The correct answer is C .
A) 11%
B) 31%
C) 35%
D) 69%
Show Answer
The correct answer is B .
Which of the following will give a precipitate with an aqueous solution of copper (I) chloride?
Options:A) But-2-ene
B) But-1-ene
C) But-2-yne
D) But-1-yne
Show Answer
The correct answer is D .
A) 0.13
B) 0.25
C) 1.25
D) 2.00
Show Answer
The correct answer is C .
A) Ionic bonding
B) Metallic bonding
C) Dative bonding
D) Covalent bonding
Show Answer
The correct answer is A .
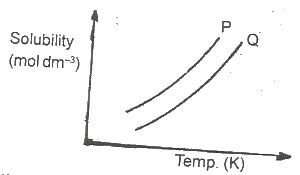
In the diagram above, the mixture of the solids P and Q can be separated by
Options:A) distillation
B) fractional distillation
C) crystallization
D) fractional crystallization
Show Answer
The correct answer is C .
What volume of 0.100M sodium trioxonitrate (V) solution contains 5g of solute.
[Na = 23, N = 14, O = 16]
Options:A) 1.7 litres
B) 0.420 litres
C) 0.588 litres
D) 2.35 litres
Show Answer
The correct answer is C .
A) 2NO(g) + O2(g) → 2NO2(g)
B) NO(g) + Cl2(g) → NOCl2(l)
C) FeSO4(aq) + NO(g) → Fe(NO)SO4(s)
D) NO(g) + Br2(l) → NOBr2(l)
Show Answer
The correct answer is A .
A)
B)
C)
D)
Show Answer
The correct answer is A .
N2O4(g) ↔ 2NO2(g) ΔH = +ve
In the reaction above, an increase in temperature will
Options:A) increase the value of the equilibrium constant
B) decreases the value of the equilibrium constant
C) increase in the reactant production
D) shift the equilibrium to the left
Show Answer
The correct answer is A .