A) SO2 and NH3
B) CO2 and H2
C) NO2 and SO3
D) SO3 and NO
E) CO and H2
Show Answer
The correct answer is C .
A) 60 cm3
B) 10 cm3
C) 40 cm3
D) 30 cm3
E) 70 cm3
Show Answer
The correct answer is A .
A) Na
B) K
C) Ca
D) Al
Show Answer
The correct answer is C .
NH4Cl(s) ↔ NH3(g) + HCL(g)
The reaction above can only attain equilibrium if
Options:A) a gaseous reactant is added
B) one of the products is removed
C) it is in a closed system
D) it is an open system
Show Answer
The correct answer is C .
A) 54.0g
B) 27.0g
C) 13.5g
D) 108.0g
Show Answer
The correct answer is A .
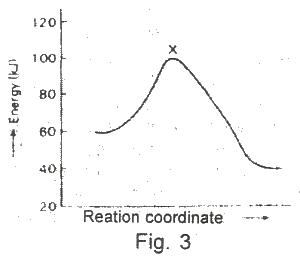
The diagram above (Fig.3) shows the energy profile for the reaction A + B + = C + D From this diagram, it is clear that the reaction is
Options:A) spontaneous
B) isothermal
C) adiabatic
D) exothermic
E) endothermic
Show Answer
The correct answer is D .
A) 16
B) 18
C) 12
D) 14
Show Answer
The correct answer is B .
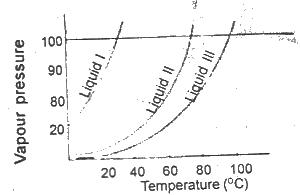
It can be deduced from the vapour pressure curves above that
Options:A) liqiud 1 has the highest boiling point
B) liquid ll has the highest boiling point
C) liquid lll has the highest boiling point
D) liquid lll has the lowest boiling point
Show Answer
The correct answer is C .
56.00cm3 of a gas at S.T.P. weighed 0.11g. What is the vapour density of the gas?
[Molar volume of a gas at S.T.P = 22.4dm3]
A) 11.00
B) 22.00
C) 33.00
D) 44.00
Show Answer
The correct answer is B .
A) Dilute ammonia
B) Benedict's solution
C) Tollen's reagent
D) Sodium hydroxide solution
Show Answer
The correct answer is C .