Which of the following mixtures is an example of a colloid?
Options:A) Milk
B) Orange juice
C) Saltwater
D) Sugar dissolved in water
Show Answer
2SO2(g) + O2(g)
In the reaction above, the concentration of 2SO3(g) can be in creased by
Options:A) decreasing the pressure
B) decreasing the temperature
C) the addition of catalyst
D) increasing the temperature
Show Answer
At 2.0 atm pressure, the volume of a gas is 4.0 L. If the pressure is reduced to 1.0 atm while keeping the temperature constant, what will be the new volume of the gas?
Options:A) 2.0 L
B) 6.0 L
C) 8.0 L
D) 4.0 L
Show Answer
220g of carbon dioxide contains ----- moles
Options:A) 5
B) 6
C) 3
D) 4
Show Answer
A) ls 22s 22P 63s23P2
B) Is22s22P6
C) ls22s22p63s2
D) ls22s22P4
Show Answer
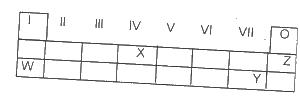
Use the table above to answer this question. The element that is likely to participate in covalent rather than ionic bonding is
Options:A) Z
B) Y
C) X
D) W
Show Answer
A) ionic
B) covalent
C) dative covalent
D) metallic
E) none of the above
Show Answer
A) 1.40 χ 102 dm3
B) 14.0 χ 102 cm3
C) 1.40 χ 10-2 dm-2
D) 14.0 χ 10-2 cm3
Show Answer
A) alkene
B) alkanal
C) alkanone
D) alkanoic acid
Show Answer
Which of the following methods is commonly used to remove suspended impurities from water?
Options:A) Fluoridation
B) Chlorination
C) Filtration
D) Distillation
Show Answer