What is the symbol used to represent an alpha particle?
Options:A) α
B) γ
C) δ
D) β
Show Answer
The correct answer is A .
If one of the following oxides is heated with hydrogen or carbon using a bunsen burner. it is not reduced to the metal, Which one is it?
Options:A) lead oxide
B) Magnesium oxide
C) Copper oxide
D) Tin oxide
Show Answer
The correct answer is B .
The Consecutive members of an alkane homologous series differ by
Options:A) CH
B) CH
C) CH
D) C
Show Answer
The correct answer is B .
A) Cl-
B) NO3-
C) SO32-
D) SO42-
Show Answer
The correct answer is C .
Proteins in acid solution undergo?
Options:A) Substitution
B) Fermentation
C) Hydrolysis
D) Polymerisation
Show Answer
The correct answer is C .
A) cacium
B) magnesium
C) tin
D) copper
Show Answer
The correct answer is C .
A) dehydration
B) esterification
C) hydrolysis
D) oxidation
Show Answer
The correct answer is C .
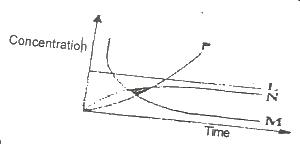
2HCI(ag) + CaCO3(s) → CaCl2(s) + CO 2(g) +H2O(1)
From the reaction above, which of the curves in the diagram represents the production of carbon (IV) oxide as dilute HCI is added?
Options:A) L
B) M
C) N
D) P
Show Answer
The correct answer is B .
A) LiCI
B) NaCl
C) CaCl2
D) MgCl2
Show Answer
The correct answer is C .
A) 400k
B) 480k
C) 550k
D) 600k
Show Answer
The correct answer is D .