A) Dehydrating
B) Becoming inert
C) Smelling
D) Catching fire
Show Answer
The correct answer is D .
A) 1 - propanol and 2 - propanol
B) 1 - propanol and 1 - propanol
C) 2 - propanol and 2 - propanone
D) 2 - propanol and 1 - propanol
Show Answer
The correct answer is A .
A) Zn
B) Fe
C) Sn
D) Cu
Show Answer
The correct answer is D .
A) excess Mg ribbon
B) excess cold water
C) very hot water
D) steam
Show Answer
The correct answer is D .
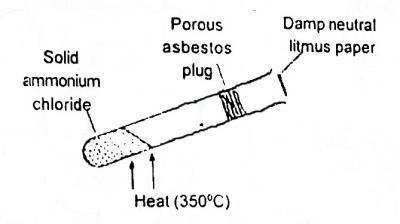
In the shown experiment (Fig. 1) the litmus paper will initially
Options:A) be bleached
B) turn green
C) turn red
D) turn blue
Show Answer
The correct answer is D .
A) Ice
B) Sulphur
C) Phosphurous
D) Iodine
E) Potassium iodie
Show Answer
The correct answer is D .
A) positive ion
B) neutral atom of a metal
C) neutral atom of a non-metal
D) negative ion
Show Answer
The correct answer is D .
A) HNO3
B) CH3COOH
C) H2SO4
D) HCl
Show Answer
The correct answer is C .
A) Equilibrium constant is very large
B) ∆H - T∆S = O
C) ∆ H > T ∆ S
D) Equilibrium constant is less than zero
Show Answer
The correct answer is A .
The fourteen elements (atomic number 90-103) after actinium are called?
Options:A) Actinoids
B) Nucleotides
C) Pre-actinum elements
D) Atomic elements
Show Answer
The correct answer is A .